What is the energy transferred from an object at a higher temperature to an object at a lower temperature?
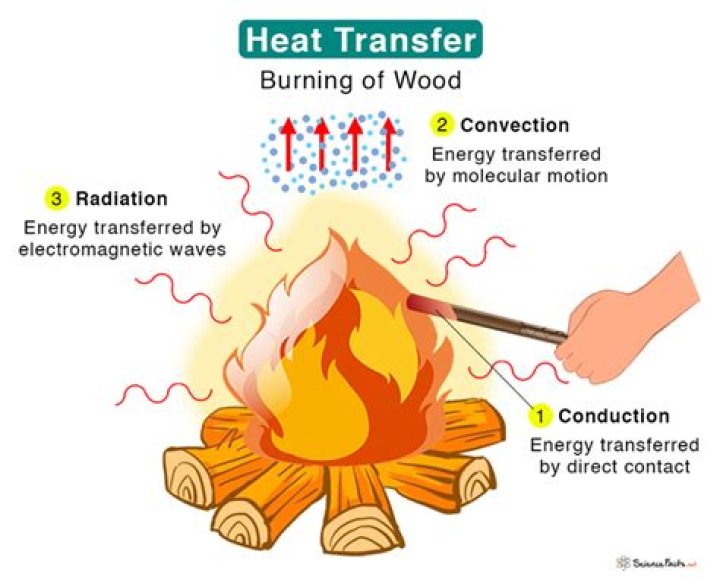
3. When objects with different temperatures are in the same environment, energy is transferred from objects at higher temperatures to objects at lower temperatures until everything is at the same temperature (thermal equilibrium). Thermal energy can be transferred three ways: by conduction, convection, or radiation.
What energy flows from a higher temperature to a lower temperature?
thermal energy
Heat: It is important to remember that heat is caused by flow of thermal energy due to differences in temperature (heat flows from object at higher temperature to object at lower temperature), transferred through conduction/convection/radiation.
What is the energy transferred from a hotter object to colder object?
Heat is the thermal energy that flows from a warmer object to a cooler object. Heat flows only one way, from warmer to cooler objects. Net heat transfer ends when two objects reach the same temperature (“thermal equilibrium”).
What is the energy transferred from a hotter object to colder one due to the difference in temperature?
Heat Transfer, Specific Heat, and Heat Capacity If two objects at different temperatures are brought in contact with each other, energy is transferred from the hotter object (that is, the object with the greater temperature) to the colder (lower temperature) object, until both objects are at the same temperature.
What are the 3 factors that determine how much thermal energy an object has?
What 3 factors influence the amount of thermal energy in an object, and how do they each affect it? Mass, temperature, and phase.
What are 5 examples of energy transfer?
Energy transfers
- A swinging pirate ship ride at a theme park. Kinetic energy is transferred into gravitational potential energy.
- A boat being accelerated by the force of the engine. The boat pushes through the water as chemical energy is transferred into kinetic energy.
- Bringing water to the boil in an electric kettle.
How do you feel when heat energy enters your body from a hotter body?
We feel cold because body is more hot than the room temperature so transfer heat energy move from higher gradient to lower so loss of heat energy causes feeling cold.
Does energy travel from high to low?
In Thermodynamics, it is said that heat transfer takes place from higher temperature to lower temperature. Things having higher temperature have higher potential energy because of more vibratory motion and other kind of motion of atoms and things at lower temperature have lower potential energy.
What is the energy transferred from a hotter object to?
Heat is the transfer of energy. During energy transfer, the energy moves from the hotter object to the colder object. This means that the hotter object will cool down and the colder object will warm up. The energy transfer will continue until both objects are at the same temperature.
What is needed for heat transfer from one object to another the two object should?
Explanation: Because in order for heat to travel onto another object, it needs to be in direct contact first.
What happens when two objects with different temperatures are in contact?
If two objects at different temperatures are brought in contact with each other, energy is transferred from the hotter to the colder object until equilibrium is reached and the bodies reach thermal equilibrium (i.e., they are at the same temperature). Heat is a form of energy, whereas temperature is not.
What are 2 factors that affect thermal energy?
An object’s thermal energy is dependent upon its temperature and mass. The higher the temperature of a given quantity of a substance, more is its thermal energy. Similarly, for the same temperature, higher mass of a substance will contain more thermal energy.
What are 2 factors that determine an object’s thermal energy?
The thermal energy of an object depends on its temperature and mass. The higher the temperature of the substance, the more thermal energy it has. For the same temperature, higher mass of a substance will have more thermal energy.
What are the 4 types of energy transfer?
There are 4 ways energy can be transferred;
- Mechanically – By the action of a force.
- Electrically – By an electrical current.
- By radiation – By Light waves or Sound waves.
- By heating – By conduction, convection or radiation.
What are 3 examples of energy transfers?
Examples of energy transfers include:
- A swinging pirate ship ride at a theme park. Kinetic energy is transferred into gravitational potential energy.
- A boat being accelerated by the force of the engine.
- Bringing water to the boil in an electric kettle.
What is the rule when it comes to hot and cold air?
In physics, the second law of thermodynamics says that heat flows naturally from an object at a higher temperature to an object at a lower temperature, and heat doesn’t flow in the opposite direction of its own accord.
What organ regulates body temperature in humans?
Our internal body temperature is regulated by a part of our brain called the hypothalamus. The hypothalamus checks our current temperature and compares it with the normal temperature of about 37°C. If our temperature is too low, the hypothalamus makes sure that the body generates and maintains heat.
What are 3 forms of energy?
Potential energy is stored energy and the energy of position.
- Chemical energy is energy stored in the bonds of atoms and molecules.
- Mechanical energy is energy stored in objects by tension.
- Nuclear energy is energy stored in the nucleus of an atom—the energy that holds the nucleus together.
What are the 4 types of heat transfer?
Various heat transfer mechanisms exist, including convection, conduction, thermal radiation, and evaporative cooling.
What happens when two objects at different temperatures are in contact?